|
Risk of atrial fibrillation or diagnosis of underlying cardiac disease Newly diagnosed hypothyroidism in a patient who is pregnant and TSH < 10 milli-international unit per liter (miU/L) Newly diagnosed hypothyroidism in a patient who is pregnant and TSH ≥ 10 milli-international unit per liter (miU/L) Primary, secondary, or tertiary hypothyroidism Recommended Synthroid starting dosage in adults For more detailed information about each dosage, see the sections below. These dosages are based on condition and body weight, which is given as micrograms per kilogram (mcg/kg). 
The dosage chart below provides an overview of Synthroid dosage recommendations for adults. The recommended Synthroid dosage for treating hypothyroidism depends on the cause of your hypothyroidism and how long since you’ve been diagnosed with the condition. Your doctor will determine the best dosage to fit your needs. However, be sure to take the dosage your doctor prescribes for you. 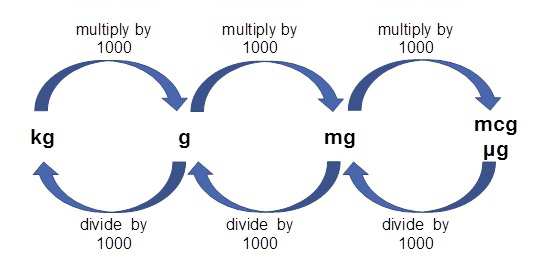
The information below describes common dosages that are typically used or recommended. See the “What factors can affect my dosage?” section below. This may also be done to reduce any side effects you’re having. Your doctor may change your dosage to make sure the drug is helping you meet your treatment goals. Your doctor will ultimately prescribe the smallest dosage that provides the desired effect. Then they may adjust your dosage as needed during treatment, by amounts of 12.5 mcg to 25 mcg. Typically, your doctor will start by prescribing a low dosage level. What are the typical dosages of Synthroid? One milligram is equal to 1,000 micrograms. Note: Synthroid comes in micrograms rather than milligrams (mg). The following strengths are also available: Synthroid is available in 25-microgram (mcg) strength intervals, as follows: 25 mcg, 50 mcg, 75 mcg, 100 mcg, 125 mcg, 150 mcg, 175 mcg, and 200 mcg. The tablets come in different colors depending on which strength they are. Synthroid comes as a tablet that’s taken by mouth. Dietary niacin can come in several forms, and the new unit accounts for tryptophan as a source of niacin.Below are details about Synthroid’s form, strengths, and typical dosages. New guidelines require that niacin be reported as Niacin Equivalents (NE). Folic acid has greater bioavailability than naturally occurring folates. New guidelines require that folates/folic acid be reported in Dietary Folate Equivalents (DFE) with mcg of folic acid in parentheses. Previously all folates and folic acid were reported as mcg folic acid. Folateĭietary folate can come in different forms naturally occurring folates and synthetic folic acid. New guidelines require that vitamin D be reported in mcg rather than IU. To convert from IU to mg alpha-tocopherol, use the 2 steps listed in the table, above. Vitamin E in food and dietary supplements can come from natural or synthetic forms. New guidelines require that Vitamin E content be determined based on the source of vitamin E and that it be reported as mg alpha-tocopherol rather than in IU. To convert from IU to RAE, use the 2 steps listed in the table, above. New labeling guidelines require reporting of vitamin A in the unit of mcg RAE, (retinol activity equivalent) in order to account for the differing bioavailability of the forms. Vitamin A has previously been reported in international units (IU). 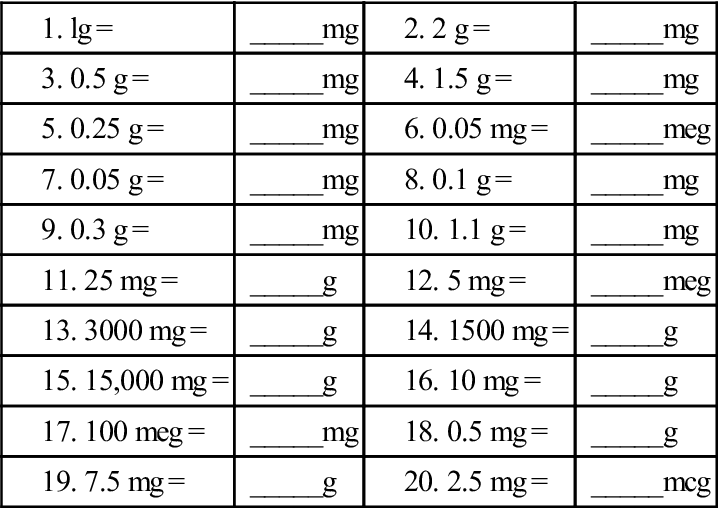
Some of these forms have greater bioavailability than others. Vitamin A in the diet can come in different forms: retinol, beta-carotene, alpha-carotene, and beta-cryptoxanthin. IU: International Unit is a measure of biological activity and is different for each substance. Labeling Units of Measure mcg: microgram. *The data in the DSID-4 are reported using the current FDA ingredient unit labeling. For more information from the FDA, click here.ġ mcg RAE = 2 mcg supplemental beta-caroteneġ IU = 0.67 mg for d-alpha-tocopherol (natural)ġ IU = 0.9 mg for dl-alpha-tocopherol (synthetic)ġ mg vitamin E (as alpha-tocopherol) label claim = 1 mg of natural α-tocopherolġ mg vitamin E (as alpha-tocopherol) label claim = 2 mg of synthetic α-tocopherol The FDA will permit manufacturers to include the amounts of nutrients in the old units in parentheses adjacent to the amounts in the new units. The changes that will affect the application of DSID results are the changes in the units used to declare vitamins and minerals on supplement labels. Voluntary compliance prior to that date is permitted, and it is expected that there will be a period of time when both types of labels will be on the market. The new regulations will be mandatory in 2019-2020. Food and Drug Administration (FDA) announced regulations that require amendments to the existing supplement facts label, which uses units and conversions based on the 1968 Recommended Daily Allowances (RDA).
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
